Worm Neurons Repaired with Genetic Engineering - 2021.01.25
Plus: A massive study explores essential genes in a famous bacterium.
☀️ Good morning
Why not start with Arthur C. Clarke’s three laws? You’ve probably heard the third:
When a distinguished but elderly scientist states that something is possible, he is almost certainly right. When he states that something is impossible, he is very probably wrong.
The only way of discovering the limits of the possible is to venture a little way past them into the impossible.
Any sufficiently advanced technology is indistinguishable from magic.
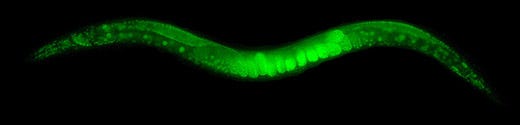
A Restored Synapse Repairs Chemotaxis
C. elegans are tiny worms, just 1mm long, that live in the soil. Under a microscope, they look like simple rods with pointed tips. But beneath their simplicity lies an amazing ability: they can sense a huge variety of olfactory and water-soluble chemicals associated with food, or nearby enemies, or other animals. Most of their nervous system, and 5% of their genes, are devoted to this chemosensing ability, according to Worm Book.
For a brilliant, open access study, published in Cell Systems, researchers at the Fred Hutchinson Cancer Research Center in Seattle delicately removed one pair of interneurons from the chemosensory circuit in C. elegans, disrupting their ability to detect and respond to environmental cues. Then, the scientists found two adjacent neuron pairs and genetically engineered them to express a gap junction protein, called connexin. This restored the chemosensory circuit in the worms. It’s a beautiful demonstration of how bioengineering can reroute neuronal connections to repair damage and tweak how information flows through the brain of an organism.
New-to-Nature Metalloenzymes
Metalloenzymes are—you guessed it—enzymes that carry around a bit of metal (in the form of an ion) that helps them catalyze chemical reactions, like reducing oxygen and fixing nitrogen. They account for about one-third of all enzymes found in nature (according to a 1996 study).
Now, there’s an easy way to create metalloenzymes that perform reactions not found in nature. For an open access study, published in Science Advances, researchers at ETH Zurich, in Switzerland, studied 400 artificial metalloenzymes, each with their own mutations, and devised a method to quickly discover which mutants could perform a desired chemical reaction. The whole process is automatable, relies on biotin-streptavidin technology, and was used to find artificial metalloenzymes that could perform “gold-catalyzed hydroamination and hydroarylation,” with 15-fold improvements in enzyme activity.
Massive Study on Essential Genes in E. coli
Every organism has genes that—if removed—would lead to a swift death. These are known as essential genes, and creating an extensive catalog of them could one day help scientists find, and create, a ‘minimal genome.’
Essential genes vary wildly across organisms, and even across closely-related strains of the same species. The model bacterium E. coli, for example, has dozens of cousins, each with their own set of essential genes. Clearly, mapping all of these genes, and figuring out how they differ across organisms and species, will require a massive experimental effort.
For a new study, published in Nature Microbiology, researchers at the Institut Pasteur, in Paris, used CRISPR interference (a technique that ‘knocks down’ the expression of genes) “to assess the importance of ~3,400 nearly ubiquitous genes in three growth conditions in 18 representative E. coli strains.” That’s a massive undertaking, and the results are worth your time: the study showed that essential genes change, depending on the environment of the host (not wholly surprising) and that some ‘core genes’ (genes that are essential across multiple species) only become essential after the organism acquires additional genes—called accessory genes—via horizontal gene transfer.
🧫 Rapid-Fire Highlights
More research & reviews worth your time
Plasmids—y’know, those loops of DNA that you use to introduce genes into a cell—may actually speed up bacterial evolution. That’s according to a new review on horizontal gene transfer, plasmids, and evolution. Nature Reviews Microbiology. Link
CRISPR-Cas9 can make cuts at inadvertent locations (called off-target effects), which is a painful byproduct when using these molecular scissors. A new protocol paper explains how to use Digenome-seq, a cell-free method to quantify off-target effects for base editors and DNA cutters. Nature Protocols. Link
This isn’t really a study about bioengineering, but it’s fascinating. A new review traces the evolutionary history of chloroplasts, and explains how they’ve been used to produce some very popular medicines. Plant Biotechnology Journal (Open Access). Link
Promoters for poplar trees: A new study designed and studied a set of “osmotic‐related and salt stress‐inducible synthetic promoters” to use in hybrid poplar plants. Wouldn’t it be awesome to design and build tree genomes from scratch? Could we make sequoias even taller? Let me know if you’re working on that. Plant Biotechnology Journal (Open Access). Link
Cas proteins can only cut where a specific ‘recognition sequence’, or PAM, happens to already exist in the genome. This review—which is great—looks at the quest for a “PAM-free nuclease.” Nature Communications (Open Access). Link
For a new study, researchers engineered E. coli bacteria to produce pyocyanin, a blue-colored toxin normally made by pathogenic Pseudomonas cells. Metabolic Engineering. Link
Prime editing can be used to make edits in a genome without cutting DNA in two. It’s a really useful technology, but requires that you design and make a distinct kind of “guide RNA”, called a prime editing guide RNA. For a new study, researchers built a software that automates this design process, called PINE-CONE. ACS Synthetic Biology (Open Access). Link
While we’re on the topic of genome editing: a new protocol describes exactly how to use base editors in mammalian cells. Nature Protocols. Link
To quickly program cells to detect environmental contaminants, a new study uses a mixture of a “DNA signal amplifier” and transducer circuit, which can “successfully detect antibiotics in nanomolar levels… suggesting great potential for monitoring of water contaminants.” ACS Synthetic Biology (Open Access). Link
How stable is a piece of RNA, floating in a bacterial cell? For a new study, researchers used synthetic biology techniques to build a large variety of RNA sequences, and then studied each of them to determine which features affected their stability. ACS Synthetic Biology. Link
For a new study, researchers created “~460,000 yeast triple mutants” to study how random sets of three genes, when disrupted, impact an organism’s health. Nature Protocols. Link
Many proteins are big; clunky even. A new study instead uses small nanobodies—essentially an antibody with just one protein domain—to control genes in mammalian cells. The nanobodies can “bind and recruit endogenous chromatin regulators to a gene.” Nature Communications (Open Access). Link
Most methods to edit a genome rely on homologous recombination, which is an inefficient process. But some bacteriophages—viruses that infect bacteria—encode proteins that can improve homologous recombination 1,000-fold. For a new study, researchers studied five different organisms to see how well these proteins work across multiple different species. Nature Chemical Biology. Link
Researchers created a bacterium that can convert tyrosine to melanin, but then also export those genes to other species. Using this “exporting” bacterium, they were able to test a simple metabolic pathway in one type of cell (Bacillus subtilis) and then transfer their design, seamlessly, to other types of cells. Metabolic Engineering. Link
Chimeric Antigen Receptor T-cells (abbreviated as CAR-T) are cells that have been genetically-engineered to target specific proteins; they can be used to fight cancer, for example. A new study introduces a “two-step positive feedback circuit” in CAR-T cells, which enables them to “sense antigen density with an ultrasensitive threshold.” bioRxiv (Open Access). Link
If you regulate both the transcription and translation of a genetic construct, you can reduce its normal expression in the cell, without impinging on its maximal expression. A new study uses this technique to create genetic systems that have “>1000-fold change in their output after induction in vivo.” bioRxiv (Open Access). Link
Pseudomonas bacteria have something called a “DNA mismatch repair system,” which is very good at repairing DNA. By manipulating these repair systems, though, researchers were able to “alter mutation rates in Pseudomonas species.” When they inhibited the system, the organism’s mutation rate increased up to 438-fold. bioRxiv (Open Access). Link
Have a great week.
Until Friday,
— Niko
Thanks for reading Cell Crunch, part of Bioeconomy.XYZ. If you enjoy this newsletter, please share it with a friend or colleague.
A version of these newsletters is also posted on Medium. Reach me with tips and feedback @NikoMcCarty or via email.


